 Subshell labels are used to write down an atom's electron configuration.Read More: Electronegativity chart Notation 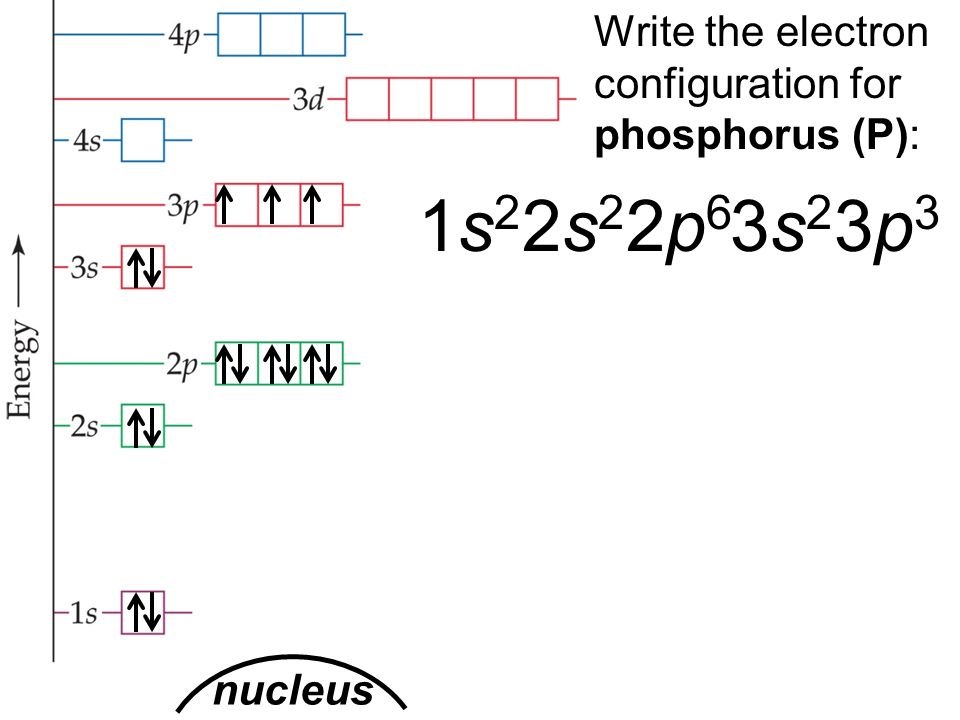
As a result, sodium's abbreviated electron configuration is 3s 1 (the electron configuration of neon is 1s 2 2s 2 2p 6, which can be abbreviated to 2s 2 2p 6).Īs a result, the 1p, 2d, and 3f orbitals do not exist because the value of the azimuthal quantum number is always less than the value of the principal quantum number.The sequence of completely filled subshells that correspond to the electronic configuration of a noble gas is replaced with the symbol of that noble gas in square brackets in the abbreviated notation.For large atomic numbers, the standard notation of electron configuration may be very long. In such cases, instead of the standard notation, an abbreviated or condensed notation may be used.The position of an element in the periodic table is determined by the quantum numbers of the last orbital filled.An electron in an atom is defined by a set of four quantum numbers (n), the most important of which defines the main energy level known as a shell.For example, the electron configuration of sodium is 1s 2 2s 2 2p 6 3s 1.Atomic electron configurations follow a standard notation in which all electrons containing atomic subshells are placed in a sequence (with the number of electrons they hold written in superscript).Electron configuration describes how electrons are distributed in its atomic subshell. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
